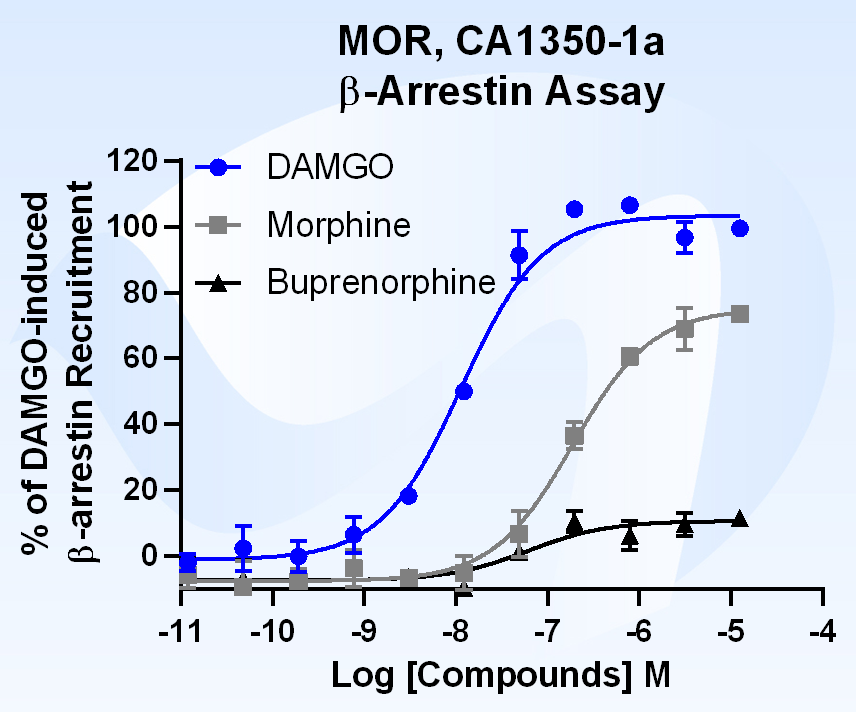
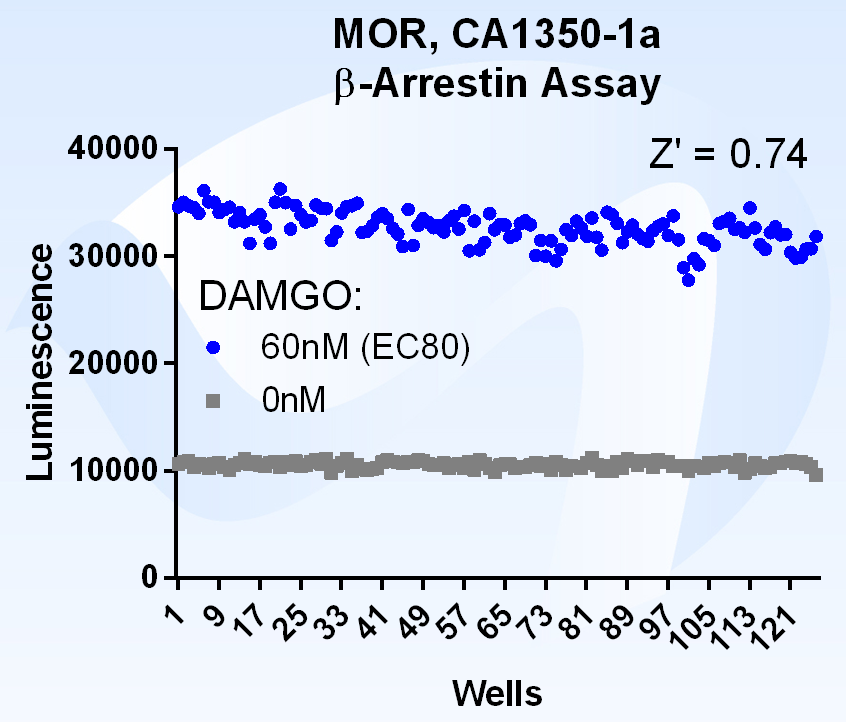
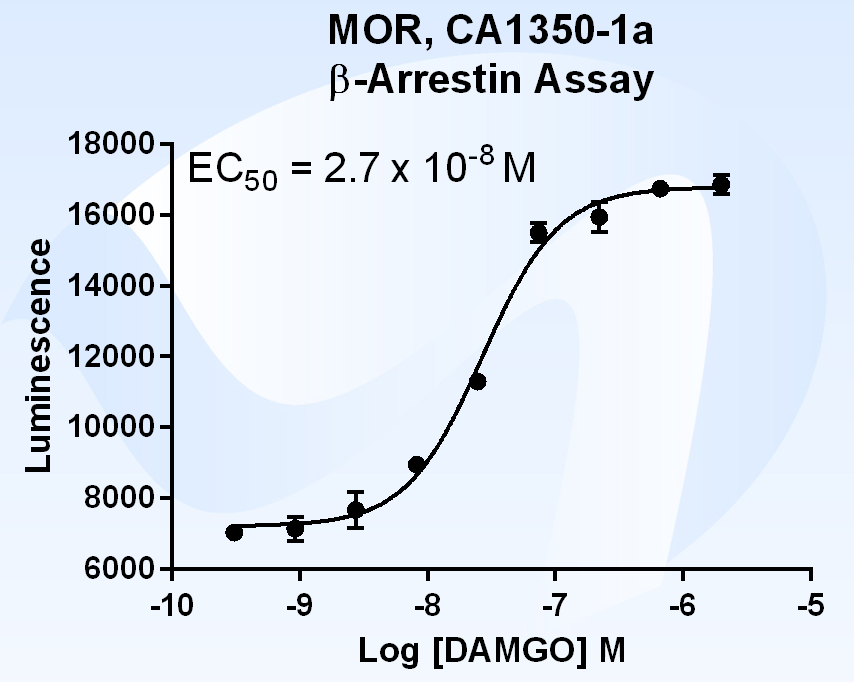
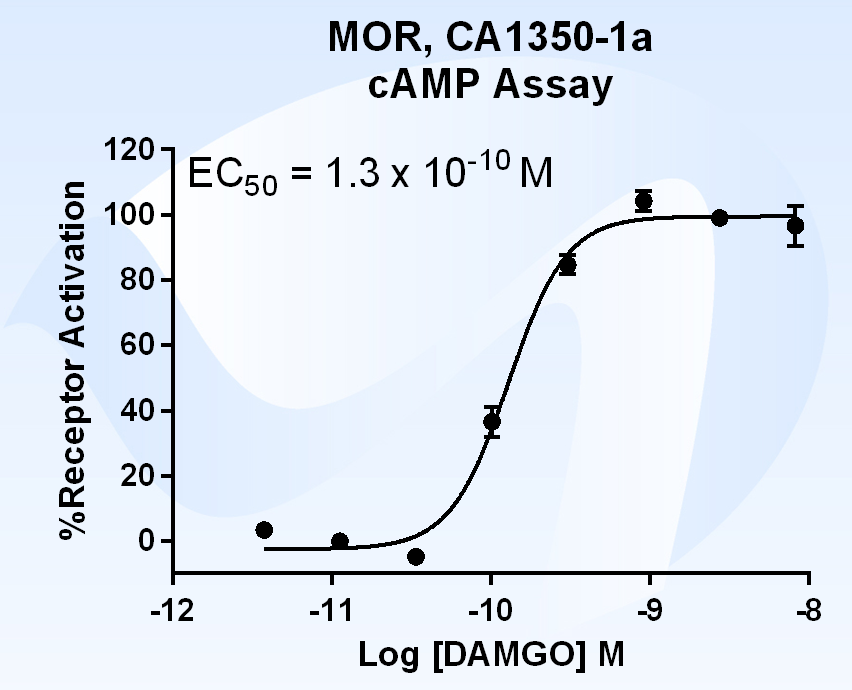
What is Beta Arrestin?
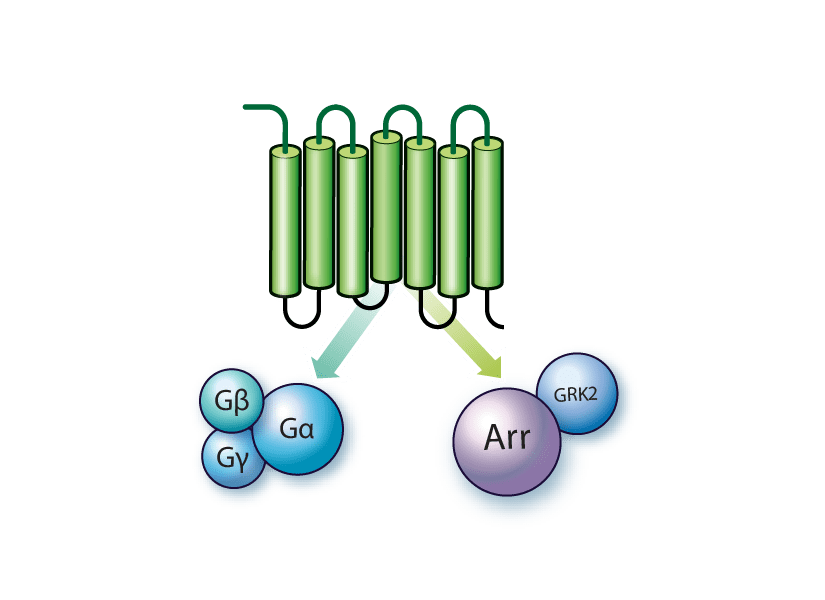
Beta-arrestin is a family of proteins involved the regulation of signaling and trafficking for GPCRs through desensitization. They can be activated by a wide range of ligands, from light-sensitive compounds to hormones and neurotransmitters. Following GPCR activation by external signals, protein activation and initiation of a signaling cascade results from its interaction with a heterotrimeric G protein. The receptor then undergoes phosphorylation by G protein-coupled receptor kinases (GRKs) to prevent prolonged or inappropriate signaling, while also marking the receptor for binding by proteins like beta-arrestin.
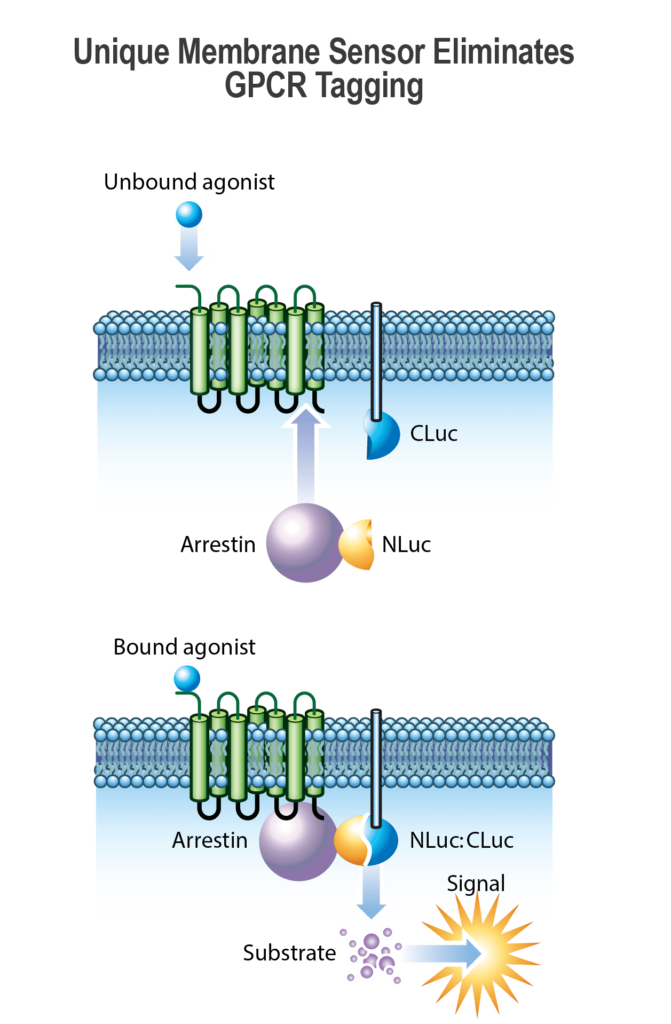
Beta-arrestin binds to the phosphorylated receptor, further inhibiting GPCR activity by preventing interactions with G proteins, while also tagging the receptor for internalization into the cell. This interaction removes the activated receptor from the cell surface, dampening the cellular response to the external signal.
Beta-arrestins are also involved in signaling pathways themselves, acting as scaffolds by organizing other signaling proteins and initiating distinct pathways independent of G proteins. There are also other forms of arrestins, including visual arrestins and cone arrestins, which are specifically involved in the rapid and highly regulated response of photoreceptor cells to light.
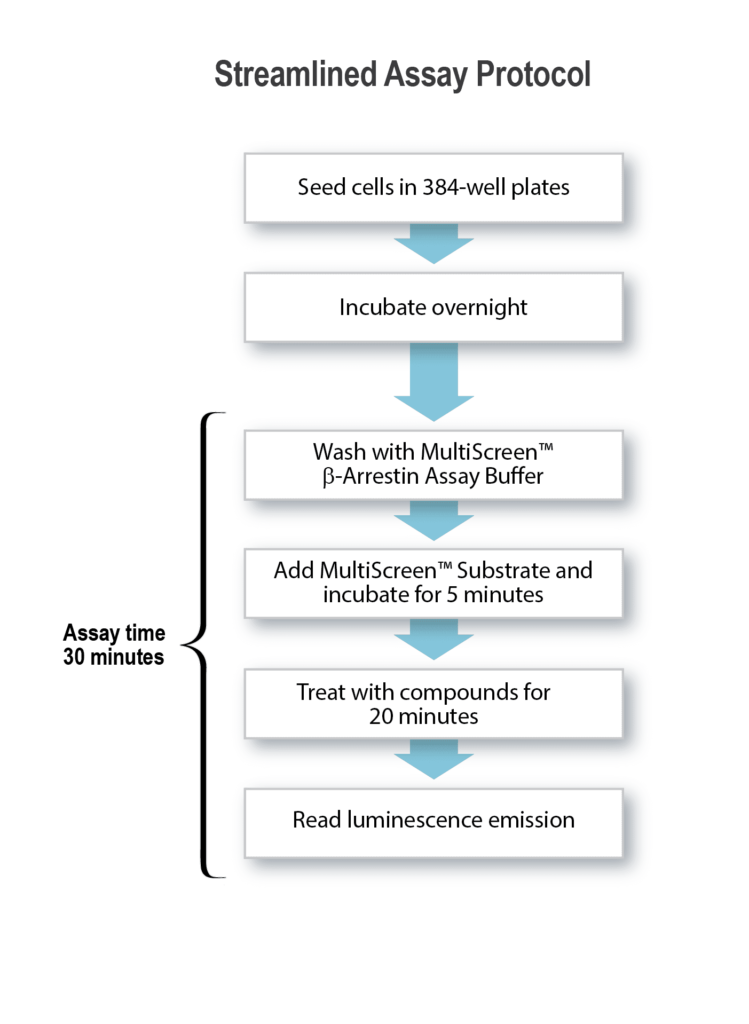
The study of beta-arrestins in live cells is essential in understanding their interactions with GPCRs, providing deeper insights into cellular communication and protein networks, and underscoring their potential as targets for therapeutic intervention in a variety of diseases where GPCR activity is dysregulated.
The bifurcation of GPCR signal transduction between G proteins and arrestins has attracted major interest in academic and pharmaceutical research on target-related pathogenesis in recent years. More importantly, possibilities of designing compounds that preferentially activate or inhibit specific GPCR signal transduction pathways have brought new hope for finding efficacious therapies without harmful side effects to the drug discovery community.