Targeting Allosteric Modulation and Cell Signaling Bias in GPCR Drug Discovery

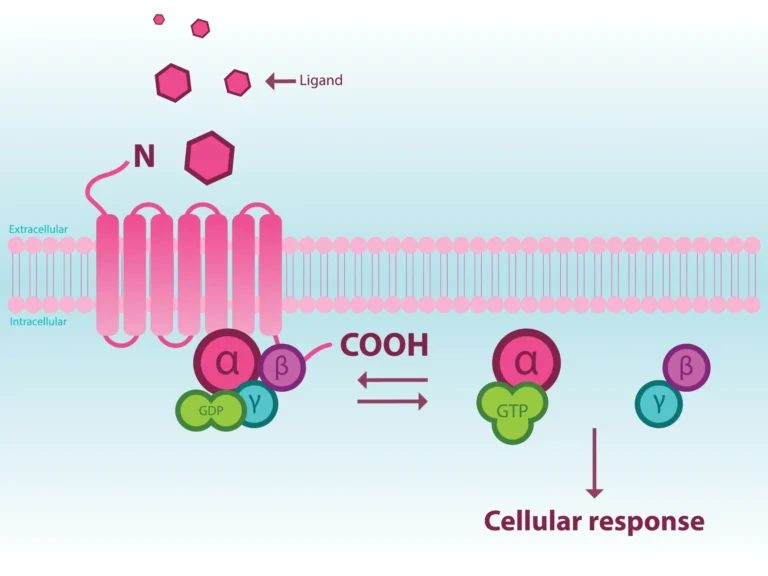
G-Protein Coupled Receptors (GPCRs) are one of the most pharmacologically successful drug targets. 35% of all drugs target this protein superfamily1. GPCR-targeted therapeutics continues to be successful and in fact, 19% of newly approved drugs target GPCRs2. The early generation of small molecule drugs targeted either orthosteric site agonist activity or antagonist activity of these receptors’ G-protein cell signaling pathway. In recent years, we have discovered more about GPCRs including crystal structures, improvements in computer-aided rational drug design, differences in the cell signaling pathways and as well as how these receptors dimerize.
Targeting Cell Signaling Bias
The increase in understanding of GPCR signaling pharmacology comes about in part from better and more sensitive screening assays. From these tools we have learned that GPCRs are much more complicated than had been believed and hence much more interesting and potentially more valuable as drug discovery targets. Evidence shows that GPCRs signal through beta-arrestin in addition to G-proteins and that ligands can trigger biased cell signaling in one direction or the other3.
This can have a therapeutic advantage such as the case of TRV-120023 in a heart failure indication. This selective molecule against the Angiotensin-1 receptor (AT1R) can not only reduce blood pressure but also positively affect heart failure. Other ARBs (angiotensin receptor blockers) did not show this effect. Why? The reason appears that TRV-120023 can block the effect of endogenous Angiotensin II while promoting cardiac contractility. Unlike its competitor ARB, Losartin, TRV-120023 activates MAPK and AKT signaling through the beta-arrestin cell signaling pathway4. This type of signaling bias has obvious therapeutic ramifications.
As a result, it appears that an important strategy would be to identify molecules that have selective patterns of cell signaling to see how they affect the desired pharmacology. This has the advantage of providing a molecule that may be efficacious toward the disease with reduced side effects due to the alternate signaling pathways.

What is Allosteric Modulation?
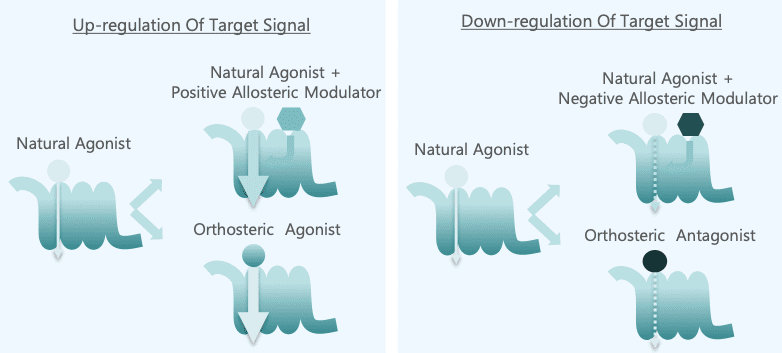
An allosteric modulator binds to an allosteric site on a protein, such as a receptor or enzyme, distinct from the active site, which alters the activity of the protein. This modulation of enzyme and receptor function differs from that of traditional agonists and antagonists. A positive allosteric modulator will enhance the affinity and/or efficacy of a receptor for its endogenous ligand or agonist, and increase the catalytic activity for enzymes. A negative allosteric modulator will reduce the affinity and/or efficacy of the receptor, and decrease the catalytic activity for enzymes.
Targeting Allosteric Modulation
An additional strategy is to identify an allosteric modulator of GPCR’s3, 5. Positive allosteric molecules have an advantage that they only work in the presence of the native ligand thus amplifying the ligand’s effect while having no activity on its own.
Demonstration of potential therapeutic benefits of allosteric modulation and liabilities of screening strategies is seen with mGLUR56. In this case, allosteric modulators of mGLUR5 were identified using a cell line transfected with an abundant amount of mGLUR5 receptors. These compounds were shown to have a liability of causing seizures in animals. However, when retested in a lower expressing cell line and native cells, it was possible to differentiate compounds that were allosteric agonists from those that were a positive allosteric modulator. The modulators no longer demonstrated seizure liability while the effectiveness against the targeted therapeutic indication, schizophrenia remained. Negative modulators are also relevant against receptors that have native constitutive activity.
A critical necessity for taking advantage of the multiple cell signaling paradigms of GPCR’s is to have the ability to measure them in robust functional assays in cells with the correct receptor expression and across appropriate orthologs to detect selectivity.
We have the capacity to measure and evaluate cell signaling bias as well as to test for positive and negative allosteric modulation. We have also retained the ability to measure traditional GPCR features such as receptor ligand binding and GTPS assay. We leverage our expertise by providing off-the-shelf products such as stable cell lines with validated assays, custom cell line generation with specific receptor expression profiles and assay development with the target of your choosing.
With our help, we can move your programs rapidly from hit identification to hit-to-lead. Moreover, we can continue working with you to optimize your compounds through SAR lead optimization. Our SAR primary screen offers unparalleled next-day turnaround. We also support secondary and tertiary screens in orthologs/paralogs GPCRs, primary cells and GPCR safety liability panels specifically designed for your program.
We have achieved a 100% return customer rate over our last decade in business and pride ourselves on excellence. Let us be your GPCR drug discovery arm.
FAQs
Orthosteric modulation is the process of a drug or ligand binding to the active site of a protein, also known as the primary site, modifying the protein behavior in response. The active site is where endogenous ligands will naturally bind to the protein. Allosteric modulation, on the other hand, involves binding on a site different from the active site. This modifies the protein’s shape, in turn changing its function.
Positive allosteric modulators and agonists share the ability to enhance receptor activity, but through different mechanisms. Agonists directly activate receptors through orthosteric modulation. Positive allosteric modulators, on the other hand, bind to a non-orthosteric site, enhancing any receptor activity in response to an endogenous ligand or agonist. Unlike agonists, allosteric modulation does not involve directly activating the receptor itself.
Allosteric modulation plays a key role in developing novel therapeutics, and offers many benefits for drug discovery researchers. For example, they can be used to adjust receptor activity in response to endogenous ligands with a high level of granularity, avoiding overstimulation or complete inhibition of receptor signals. They can also synergistically work with endogenous ligand signaling to offer greater therapeutic effects.
Allosteric modulators change the shape of the enzyme they bind to, which changes the activity of the enzyme itself. With positive allosteric modulators, the enzyme’s structure is changed in a way that makes its orthosteric site more accessible for binding, increasing enzyme activity. In the case of negative allosteric modulators, the enzyme’s structure is changed to make the orthosteric site less accessible for binding, inhibiting enzyme activity.
Common examples of positive allosteric modulators include benzodiazepines for anxiety and seizure disorders (GABA receptor), buspirone for anxiety disorders (serotonin receptor), and cinacalcet for hyperparathyroidism (CaSR receptor). Examples of negative allosteric modulators include memantine for Alzheimer’s disease (NMDA receptor) and maraviroc for HIV treatment (CCR5 receptor). Allosteric modulators can provide highly targeted therapeutics when compared to traditional drugs due to their enzyme-specific modulation.
References:
- Sriram Kand Insel A, GPCRs as targets for approved drugs: How many targets and how many drugs? Molecular pharmacology Jan 3, 2018, mol.117.111062; DOI: https://doi.org/10.1124/mol.117.111062
- Khoury E et al, Allosteric and biased g protein-coupled receptor signaling regulation: potentials for new therapeutics. Front Endocrinol (Lausanne). 2014 May 8;5:68. doi: 10.3389/fendo.2014.00068. eCollection 2014.
- Kim KS et al, β-Arrestin-biased AT1R stimulation promotes cell survival during acute cardiac injury. 2012 Oct 15;303(8): H1001-10. doi: 10.1152/ajpheart.00475.2012. Epub 2012 Aug 10.
- Wootten D et al, Emerging paradigms in GPCR allostery: implications for drug discovery, Nat Rev Drug Discov. 2013 Aug;12(8):630-44. doi: 10.1038/nrd4052.
- Rook JM et al, Unique signaling profiles of positive allosteric modulators of metabotropic glutamate receptor subtype 5 determine differences in in vivo activity. Biol Psychiatry. 2013 Mar 15;73(6):501-9. doi: 10.1016/j.biopsych.2012.09.012. Epub 2012 Nov 7