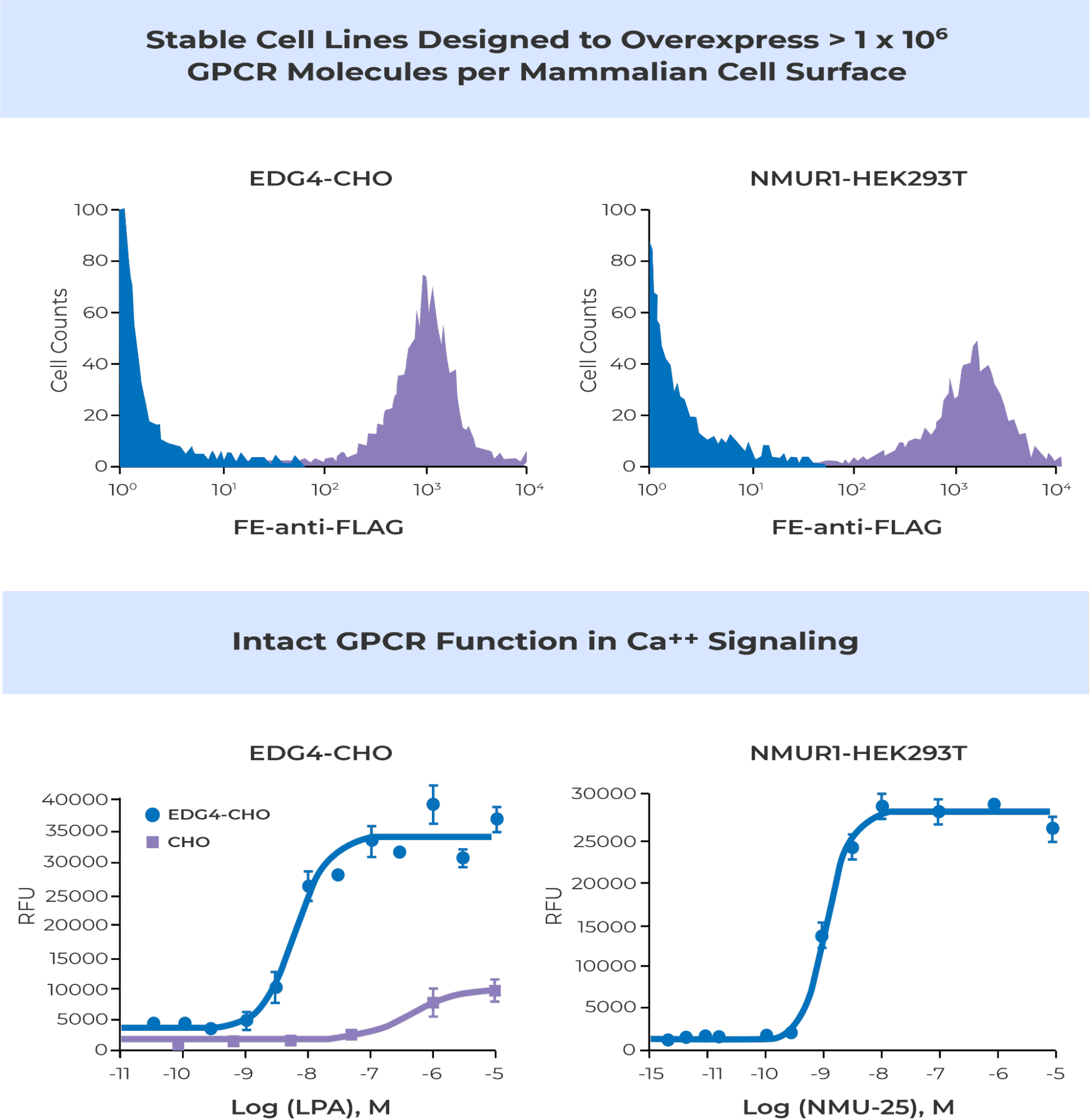
Multi-transmembrane proteins are far more challenging to express in mammalian cells than cytosolic or single-pass transmembrane proteins. G protein-coupled receptors (GPCRs) are seven-transmembrane spanners. A key question to ask when engineering mammalian cells to express GPCRs is “What’s the right expression level?” The answer is always “It depends!”
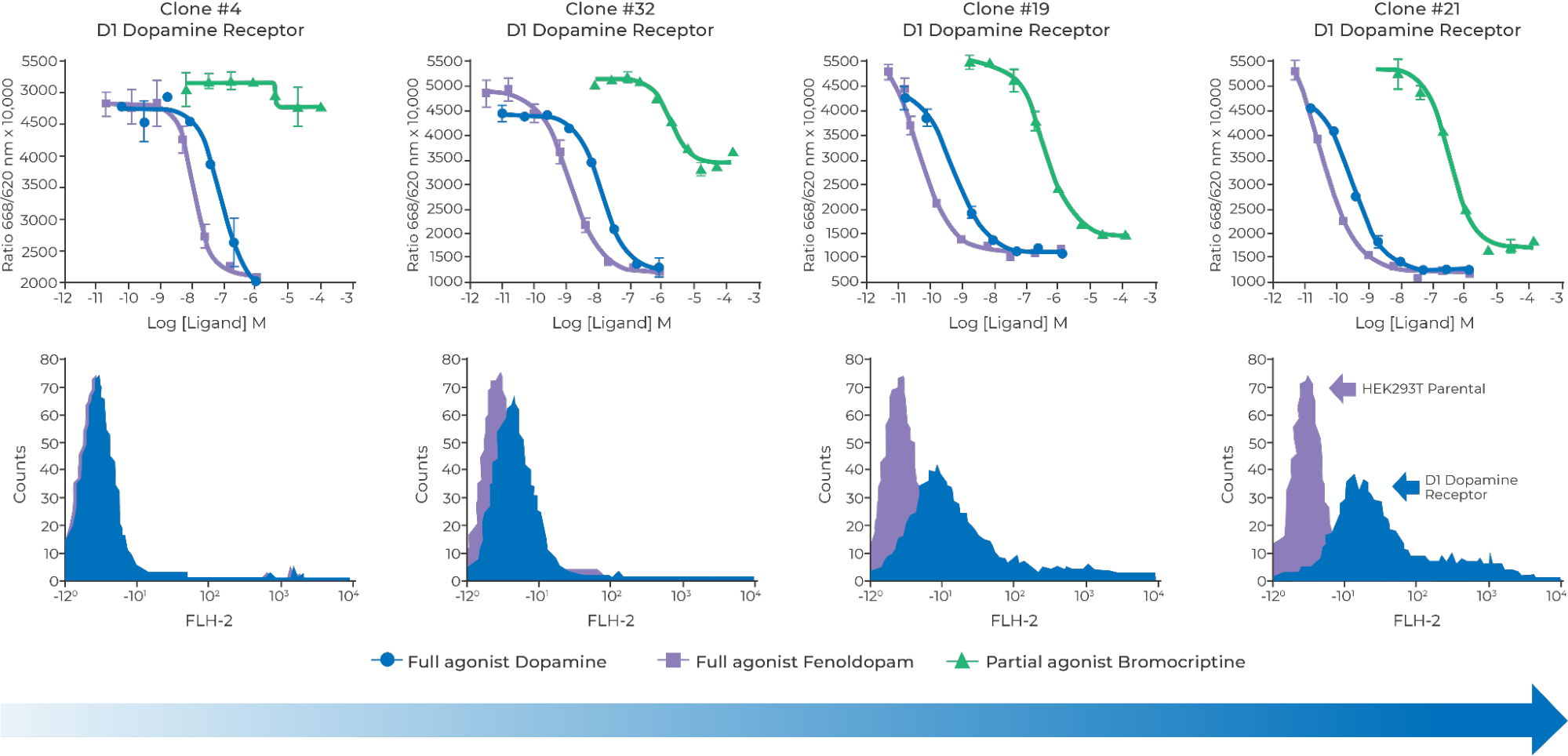
More specifically, for immunizing animals to generate GPCR antibodies or screening GPCR antibodies, small molecules or peptide binders, high cell surface expression is generally beneficial. In contrast, it is far more complex when it comes to developing stable cell lines for the purpose of cell-based assays. Depending on the modality of the high throughput screen and the pharmacology of each GPCR, different expression levels may be appropriate for different GPCRs and different screens. There isn’t a one size fits all.
To meet the needs of various drug discovery and research interests, Multispan developed its own patented proprietary vectors, designed for robust surface expression of GPCRs and other multi-transmembrane proteins in mammalian cells. Combined with Multispan’s in-depth know-how in cell engineering and single cell cloning, these proprietary vectors are part of an innovative, powerful and versatile GPCR expression technology platform.