2020 was a banner year for CXCR7. Two research studies and several great review articles were published that delve into the biased nature of CXCR7 signaling, its role in cancer, and the development of CXCR7-selective inhibitors.
- CXCR7: A β-arrestin-biased Receptor That Potentiates Cell Migration and Recruits β-arrestin2 Exclusively Through Gβγ Subunits and GRK2
- The Role of the CXCL12/CXCR4/CXCR7 Chemokine Axis in Cancer
- Advances in CXCR7 Modulators
- Discovery of the Potent, Selective, Orally Available CXCR7 Antagonist ACT-1004-1239
Atypical chemokine receptors (ACKRs) are a group of G protein-coupled receptors (GPCRs) that do not stimulate typical Gα-mediated signaling pathways upon ligand binding, and thus have been thought to act as decoy receptors to modulate chemokine responses. But more in-depth analyses, particularly research focused on chemokine receptor 7 (CXCR7), is changing that thinking.
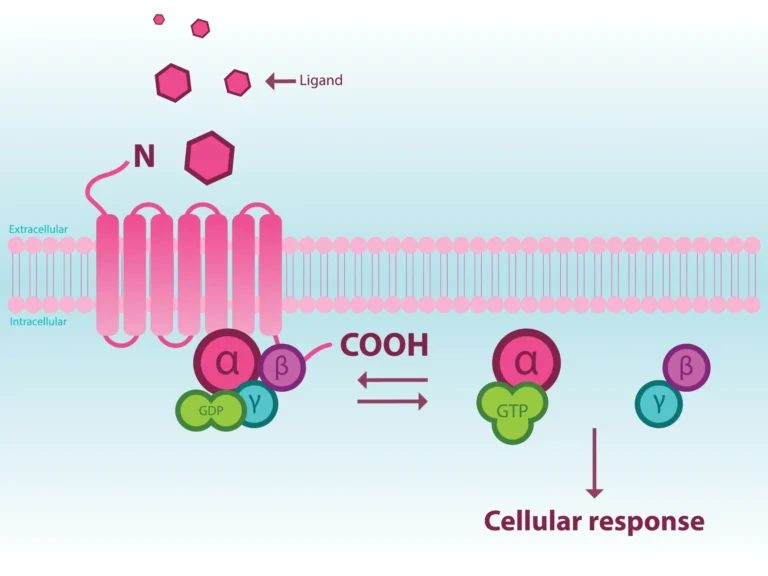
CXCR7, also known as ACKR3, binds interferon-inducible T cell α chemoattractant (I-TAC or CXCL11) and stromal cell-derived factor-1 (SDF-1 or CXCL12), which leads to recruitment of β-arrestin. SDF-1α is also a ligand for CXCR4, a conventional chemokine receptor, that unlike CXCR7, activates Gα-mediated signaling as well as G-protein receptor kinase (GRK)-mediated β-arrestin recruitment.
The SDF-1α/CXCR4/CXCR7 axis is important in many disease pathologies including neurological, cardiovascular, and inflammatory conditions, as well as tumor development and metastasis. In December of 2020, an in-depth review in Frontiers of Pharmacology by Shi et al. was published that details what is known about SDF-1α-induced CXCR4 signaling pathways and the role of these pathways in mediating cellular outcomes like chemotaxis, proliferation, survival and differentiation and their relationship to cancer (1). It further discusses SDF-1α-induced CXCR7 signaling and highlights how little is known about its mechanism of signaling relative to CXCR4.
Because of its importance in disease pathologies the SDF-1α/CXCR4/CXCR7 axis represents a promising target of therapeutic intervention. While a few drugs that target this axis have been approved for clinical use to-date, their development is challenged by CXCR4/CXCR7 crosstalk and the lack of detailed knowledge of CXCR7 signaling. In early 2020, an article entitled ‘Advances in CXCR7 Modulators’ was published that reviews the role of CXCR7 in disease as well as avenues of CXCR7 modulation that are in development again highlighting the intense interest of CXCR7 as a drug target (2). Additionally, in December of 2020, Richard-Bildstein et al. reported in the Journal of Medicinal Chemistry the discovery and development of ACT-1004-1239, a compound with high CXCR7 antagonistic activities and balanced ADMET properties (3).
CXCR7, like CXCR4, interacts with β-arrestin2 upon SDF-1α binding, however, mediators for β-arrestin2 recruitment remained unclear. A recent study published in Cell & Bioscience from Nguyen et al. looked to characterize SDF-1α-dependent signaling via CXCR4 and CXCR7 using an array of methods including structural complementation assays, CRISPR gene deletion, and selective signaling inhibitors (4).
The authors found that while SDF-1α binding to CXCR4 induced Gαi activation and β-arrestin2 recruitment through C-terminal phosphorylation by both GRK2 and GRK5, binding to CXCR7 led to activation of GRK2 via Gβγ subunits with subsequent β-arrestin2 recruitment. Although different divergent signaling pathways were used, stimulation of both CXCR4 and CXCR7 via SDF-1α binding was required for cell migration. Cumulatively their data demonstrate that CXCR7 signaling following SDF-1α engagement involves a Gα-independent, GRK2/3-mediated mechanism of β-arrestin2 recruitment and that CXCR7 is not simply a chemokine decoy but plays an active role in mediating cell migration.
In the banner year of 2020, tools like CRISPR engineered cell lines, β-arrestin recruitment assays, and GRK phosphorylation assays made headway in understanding CXCR7 signaling and advanced it as a potential therapeutic target. Multispan doesn’t want cell line generation, CRISPR engineering, or high throughput signaling assays to hold back your chemokine research. We have extensive experience with GPCRs and a line of 50 clonally-derived chemokine receptor cell lines spanning all 20 chemokine receptors – including a NEW CXCR7 β-arrestin2 Assay for ‘tag-less’ evaluation of β-arrestin recruitment. Let our cell lines, signaling assays, and cell engineering and assay services propel your drug development & research initiatives.
Other Scientific Insights from Multispan:
- Don’t let GPCR tagging bias your results. Find out how to unbias your GPCR signaling research with novel β-arrestin recruitment assays.
- Harness the power of CRISPR Engineering. Find out how Multispan solutions ensure production of high quality CRISPR tools.
- Not all cell lines are created equal. Find out why Multispan cell lines outperform.
- The Role of the CXCL12/CXCR4/CXCR7 Chemokine Axis in Cancer. Front. Pharmacol. 11:574667 (2020). doi: 10.3389/fphar.2020.574667.
- Advances in CXCR7 Modulators. https://www.mdpi.com/1424-8247/13/2/33/htm
- Discovery of the Potent, Selective, Orally Available CXCR7 Antagonist ACT-1004-1239.J Med Chem. (2020); 63(24):15864-15882. doi: 10.1021/acs.jmedchem.0c01588.
- CXCR7: A β-arrestin-biased Receptor That Potentiates Cell Migration and Recruits β-arrestin2 Exclusively Through Gβγ Subunits and GRK2. Cell Biosci. (2020) 10, 134 doi.org/10.1186/s13578-020-00497-x.
Email info@multispaninc.com
Let’s discuss how MULTISCREEN™ cell membranes can uniquely empower your program.