GPCR Membrane Ligand Binding Assay Development: The Critical Role of Cell Membranes

Explore the many types of ligand binding assays using GPCR membranes and the importance of high-quality membranes for assay development.
A ligand is a substance that forms a complex with a biomolecule, often expressed in the cellular membranes such as G-protein coupled receptors (GPCRs). The introduction of Membrane Ligand binding assays and High-Throughput Screening (HTS) has revolutionized medicine. Ligand binding allows us to understand how a ligand interact with a target protein and to measure the force (affinity) and speed (kinetics) of the interactions, while HTS helps automate research and reduce development time.
If you have questions about ligand binding using cell membranes, this article is for you. Read on to learn more.
Biochemical Ligand
A biochemical ligand forms complex with its protein targets. For example, protein-ligand binding targets produce signals by binding to target proteins. The binding often leads to a conformation change of the target protein. Binding can occur because of Van der Waals forces, ionic bonds, and hydrogen bonds. It is uncommon for ligand bonds to be irreversible. Affinity is the measurement of the strength or tendency of a ligand binding effect. Ligands can be any of the following: antagonists (inhibitors); agonists (activators); positive allosteric modulators (PAMs) and negative allosteric modulators (NAMs).
G-Protein Coupled Receptors
GPCRs are a large family of cell surface receptors and G-proteins are special kind of proteins found inside the cell cytoplasm. GPCR proteins bind ligands outside of cell membranes, which in turn trigger their binding to G-proteins inside a cell and subsequent cell signaling cascade.
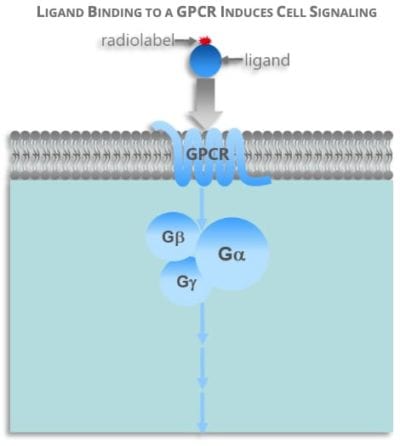
GPCRs are in almost all organisms. Mammals, invertebrates, and microorganisms all have them. You can find almost 1,000 different types of GCPRs in humans alone.
GPCRs respond to a wide range of ligands including:
- Light
- Peptides
- Lipids
- Sugars
- Other Proteins and Hormones
Ligand Binding Assays Use Cell Membranes
Because GPCRs are so prolific in the human genome, most modern drugs use them GPCRs as targets. Changes in cell membrane and membrane itself, control cell signaling and ligand binding in GPCRs. This is important pharmacology because it gives drugs a way to identify, target, and affect specific proteins.
Radioligand and Radioligand Binding Assay
Radioligands are radioactive isotope-labeled compounds. Radioligand binding assays provide sensitive and quantitative information about GPCR expression and affinity, as wells kinetics. Conventionally, it has been essential for drug structure-activity relationship (SAR) and structural–selectivity relationship (SLR) in drug discovery and basic GPCR research. There are three basic radioligand binding assay protocols: saturation, indirect (competition, displacement, or modulation), and kinetic binding. They are used to assess GPCR expression (Bmax), equilibrium dissociation constants for radioligands (Kd) and nonradioactive ligands (Ki), and association (Kon) and dissociation (Koff) rates. They are also used to distinguish competitive and allosteric mechanisms of GPCR-ligand interactions. Nonspecific radioligand binding are determined by appropriate choices of reaction conditions, for example, by inclusion of large amount of competitive cold ligand.
Special attention is required under certain circumstance. One example is radioligand depletion (bound radioactivity >10% of total radioligand), which compromises accuracy of Kd and Ki measurements. In this situation, various receptor concentrations need to be tested to ensure bound radioactivity <10% and if necessary, choose a different radioligand. Accurate Kd and Ki values in saturation and indirect binding assays depend on binding equilibrium. Extended incubation times or increased incubation temperature may be required to achieve binding equilibrium for ligands with slow kinetics.
Radioligand binding has been the gold standard for GPCR binding assays. High quality GPCR membrane is essential for GPCR radioligand binding assays.
In recent years, new methods have been developed but the knowledge learnt from radioligand binding has been the guiding principle for all new methods.
Fluorescently-Labeled Ligand
Fluorescently-labeled ligand opened up a such new frontier. The reason GPCRs react to so many stimuli in so many ways is still a mystery. Yet recent research shows that fluorescence spectroscopy may help lift the veil. Through fluorescence imaging, we’ve learned that a single cell membrane can react in a variety of ways depending on the catalyst.
Fluorescently-labeled GPCR ligands have the following advantages:
- Locate tissue and cell receptors
- Replace radioligands for simple binding assays
- Understand ligand-receptor formations and mechanics
GPCR ligands need modifications for them to become fluorescent, and since fluorescent labels are usually much larger than radioisotope, they often threaten to alter the ligand’s activities. For this reason, fluorescent ligands are not a common replacement for radioligands. Despite this drawback and decades of research, there is much to learn from these studies.
Surface Plasmon Resonance (SPR)
Surface Plasmon Resonance (SPR) is another new tool used in biochemical and biotechnological fields that’s only two decades old. It is advantageous because it does not require labeling the ligand. It uses evanescent waves to evoke a response from cell membranes with GPCRs. SPR is sensitive enough to provide a plethora of real-time data. Combine this with SPR imaging (SPRi) and you’re able to see where those readings originate from.
Additionally, SPR biosensors don’t react to the omnipresent electromagnetic interference in the modern age. This advancement has seen use in food processing, diagnostic analysis, and environmental monitoring.
High-throughput screening (HTS)
High-throughput screening (HTS) is a type of experimentation used in biology and chemistry.
HTS uses a variety of methods and technologies to enable efficient research including:
- Data processing and control software
- Robotics
- Liquid handling devices
- Sensitive detectors
Through HTS, researchers can conduct millions of tests in a short amount of time. This analysis identifies active compounds, antibodies, and genes that control biomolecular pathways. The scientists can better understand the role of specific receptors like GPCRs for reaction to certain stimuli or the lack thereof.
Armed with this information, scientists can also develop more effective drugs.
Vast improvements in HTS assay development have been made in the last two years. Key advancements in the following areas have made this significant progress possible:
- Enhanced liquid handling and Expanded automation
- Increased communications between scientists
- Better access to reagents, proteins, and engineered cell lines
- More experience with HTS and target cells
- Advances in technology and approaches
SPA (Scintillation Proximity Assay)
SPA (Scintillation Proximity Assay) is a plate based high throughput radioligand binding assay. SPAs are enabled by the diversity of radiolabeled molecules and affinity tags that are commercially available. These synthetic radiotracers allow for minimal disturbance of the natural binding interaction. It has been widely used in GPCR drug discovery research, and is a powerful tool for high-throughput screening (HTS) because they can measure the binding of very diverse classes of drug targets. it is possible to build a large variety of miniaturized, high-throughput assays and screen millions of compounds.
84% of HTS labs now report their assay development process takes six months or less. If you need help customizing your process, ask your HTS vendor for advice. Many vendors confirm their assay kits and commercial reagents. This specialized experience can come in handy when you need unique solutions.
Multispan is happy to help you have any questions about improving your processes and beat timelines. We’ll get back to you within 24 hours of receiving your email.
Make Better Medicine With Multispan
High-Quality Membranes in Binding Assays are essential to produce solid, reproducible results in HTS. Low-caliber membranes may respond in different ways from what would reflect your target’s true native pharmacology. This not only could cause inaccurate test results but also lead to added time spent on HTS.
Multispan offers SIGNATURE MULTISCREEN™ GPCR membrane solutions for your HTS needs. Using these membranes for ligand binding offers the following distinguished benefits:
- High target specific activity.
- Reduced lot-to-lot variation by generating from MULTISCREEN™ stable cell lines.
- Each batch QC’ed by cell surface expression or function.
- Available in both human and ortholog GPCR targets.
Your success is Multispan’s priority. We can deliver membrane for your target from our over 500 well-validated MULTISCREEN™ stable cell lines or we can custom produce it for you. We encourage you to take a look at our informational posters for detailed data sets. You can also follow us on LinkedIn and we’ll keep you updated with our important developments!
Let us help you make better medicine, one assay at a time.
FAQs
Ligand binding is the process where a molecule (ligand) binds to a receptor site on a target protein. This interaction causes changes to the receptor’s shape that influence its function. This can include enhancing affinity for the ligand, increasing sensitivity, or amplifying the signal by triggering signaling pathways. Ligand binding is essential for cells to adapt to external stimuli and adequately respond by adjusting cellular response in a timely manner.
Flow cytometry can be used in measuring ligand binding in assays, but is not a ligand binding assay itself. Rather, flow cytometry can measure the amount of ligand binding through quantifying fluorescence intensity corresponding to a ligand’s fluorescent tag. This is one specific use of fluorescent labeling, as flow cytometry can also be used to analyze cell size, cell count, and other important characteristics.
A radioligand is simply a radioactively labeled ligand, and is often used to study binding characteristics of receptors, such as receptor density, distribution, or affinity in the lab. A radiotracer, on the other hand, is a radioactive compound used in a wider range of scientific and medical applications, such as imaging studies and disease diagnosis. While radioligands focus on receptor interactions at the molecular level, radiotracers are used for measuring biochemical processes in the body.
Functional assays are used to measure biological activity after binding has occurred, with a focus on a ligand interaction’s influence on targeted physiological processes. Unlike binding assays, functional assays do not assess affinity or biophysical specificity of the ligand-binding. Functional assays are essential in determining the effects of a potential therapeutic drug.
Competitive binding assays are a type of ligand binding assay, used to study the relative affinity of different molecules to the same enzyme or receptor. During this process, a labeled ligand is put into competition with a test ligand for the same receptor, and binding activity between the two is measured. These assays are particularly useful in agonist or antagonist screening, where a molecule’s potential to outcompete a well-defined labeled tool molecule for the same receptor is researched.
E-mail info@multispaninc.com
Let’s discuss how MultiScreen™ cell membranes can uniquely empower your program.