Meeting The Need For Accelerated Covid Antiviral And Vaccine Development
The rapid emergence of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) or COVID-19 has substantially impacted both human health and the global economy.
Helping Make a Difference
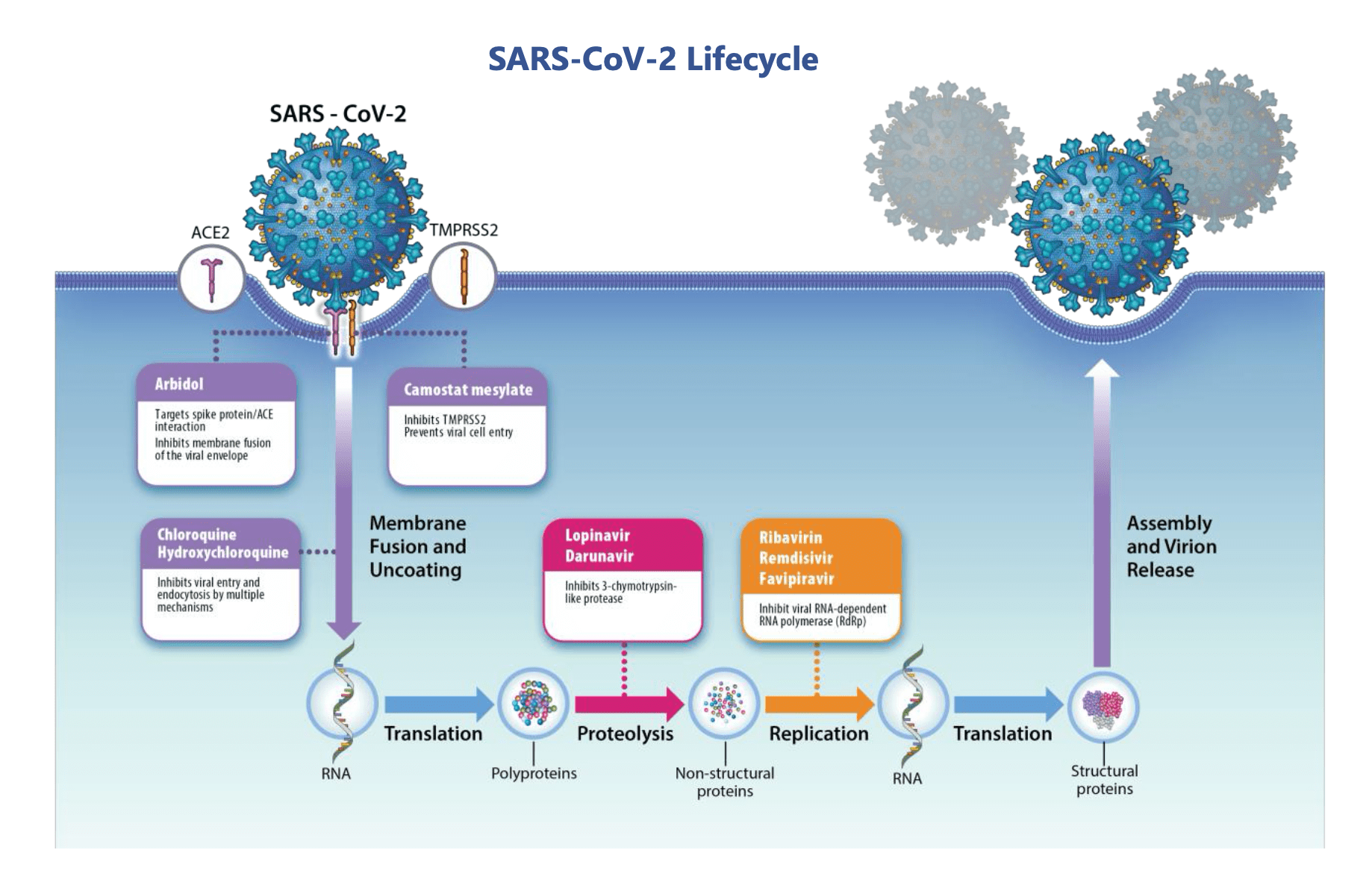
Control and eradication of SARS-Cov-2 will require advanced antivirals and/or a protective vaccine – development of both of which depends on improved knowledge of the virus pathophysiology, identification and therapeutic exploitation of the vulnerabilities of the virus, and characterization of the components of protective immunity.
A variety of assays are needed ranging from general infectivity to highly specific viral pathways including binding, entry, enzymatic activity, maturation, and packaging.