What Does CRISPR Success Look Like?
At the highest level, most all of us would say that CRISPR success looks like patients with better disease outcomes and improved quality of life. At the level of cell and gene therapies this success is derived from the efficient repair of a double stranded DNA break in the cells of interest – thus creating an efficacious therapy.
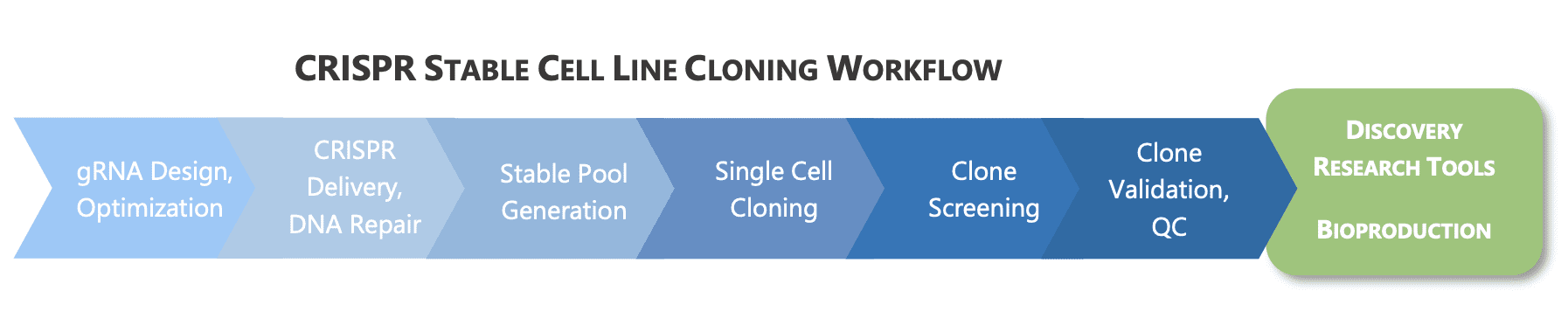
The impacts of CRISPR, however, extend beyond cell and gene therapy into the domain of small & large molecule therapeutics. CRISPR ‘success’ in these areas requires going beyond DNA repair and into the world of cell line engineering. The goal of these CRISPR initiatives is the generation of stable cells containing the genomic alteration(s) of interest that are used as tools for discovering new drugs or for the bioproduction of biologics – in the end bringing new therapies to market.
CRISPR Accessibility
CRISPR has unlimited potential to accelerate drug discovery from early target identification and candidate screening through lead optimization and pre-clinical animal modeling. In response, laboratories across the globe are looking to join the CRISPR revolution.
In theory, CRISPR is deceivingly simple – develop a target-specific guide RNA and the world of gene editing opens. Commercial companies have developed CRISPR tools and off-the-shelf products that provide wider access to this technology; however, substantial barriers remain. Many laboratories simply don’t have the breadth of knowledge or diversity of skills for successful implementation of CRISPR which can include guide RNA design and optimization, delivery of CRISPR components (transduction or transfection), bioinformatics, molecular biology, and cell culture. Ultimately, it is the ability to generate quality cell lines stably expressing the genomic alteration(s) that bring CRISPR initiatives aimed at creating tools to fruition.
Clonality Counts
In general, two options are available for stable expression of CRISPR-edited genetic modifications: generation of stable pools or clonally derived, stable cell lines.
Stable pools are substantially easier and faster to create than stable cell lines and thus can be tempting to use, but beware — clonality counts.
CRISPR generated stable pools are a heterogeneous mixture of un-engineered cells and cells containing a variety of genomic alterations. For example, stable pool variability for a CRISPR gene knockdown study may include cells with different size deletions, different effects on the expression level of the knocked down gene, and/or disruption of other off-target genes. A population of cells with heterogeneous genotypes such as these will result in mixed phenotypes which in the end can mask experimental findings.
In addition to ambiguous results, stable pools, unlike their name, are not stable. The mixture of cells continues to evolve over time as cells compete to outgrow one another. This instability and the resulting lack of reproducibility adds an additional layer of complexity in data interpretation.
While, stable pools are sufficient for some drug discovery efforts such as target discovery and validation early on, most applications benefit from a clonally derived stable cell line whose genetic alteration(s) have been well characterized and clones selected based on the desired phenotype — both important parameters for the reproducible production of high-quality data. Thus, the decision to generate stable cell lines as opposed to stable pools may be key to the power of your CRISPR initiatives.
Not All Stable Cell Lines are Created Equal - It's in the Details
Stable cell line generation remains a complex, resource intensive, and time-consuming process that many labs are unfamiliar with. Yet it is a critical factor in the success of CRISPR engineering initiatives. Thus, laboratories often turn to outsourcing.
Put simply — not all stable cell lines are created equally. It requires a high degree of expertise and rigor to establish and validate a quality, clonally derived stable cell line with precise genomic alterations. Consequently, care should be taken in choosing an outsource provider. Here are a few details to consider:
- Is the provider experienced creating and documenting the derivation of a cell line from a single cell? This can be imperative for experimental stability as well as the required provenance for use of the cell line in bioproduction.
- Do they perform stringent validation of CRISPR genomic alterations and cell line QC? While CRISPR editing is ‘targeted’, it has variable editing efficiencies and potential off-target alterations as well as deletion size variability. Care should be taken to characterize not only genomic alterations but also protein expression levels to ensure experimental findings can be interpreted.
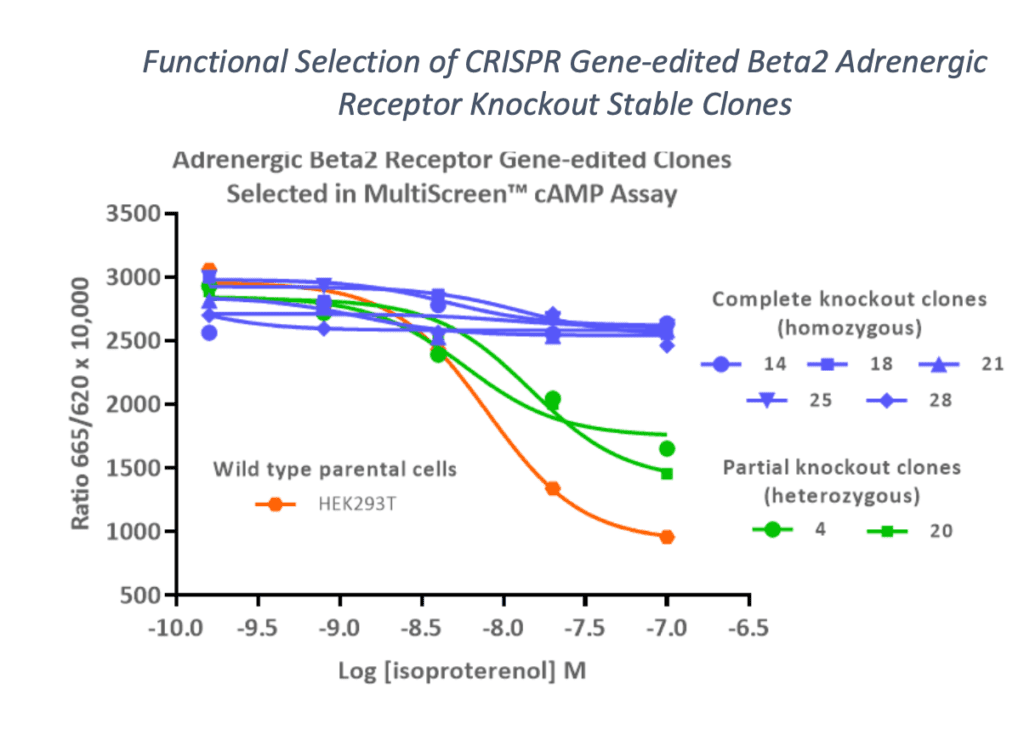
- Are they familiar with selecting clones based on functional assays? If the CRISPR-engineered cell line will be used as tools, it is highly advantageous to screen clones based on performance within these functional assays in the intended assay format, such as 384-well microplates. Additionally, it provides evidence that normal house-keeping endogenous pathways remain intact.
- Can they rapidly generate a range of CRISPR cell lines including homozygous vs. heterozygous genomic alterations, a panel of mutations, or in multiple cell lines? Having a pair or even a series of CRISPR cell lines, including wild-type or parental cells, is a helpful tool that supports a variety of applications throughout the lifecycle of a project.
- Do they offer other CRISPR assay services? Often you may be looking to accelerate drug discovery by outsourcing other areas of development such as cell expansion or banking, assay development, high throughput screening, lead optimization, or structure activity (SAR) testing. Identify a provider that fully support your needs.
Stable cell line generation can take months, but it is important to get the details right. Take the time to find a partner whose expertise you can count on with an unmatched track record of success to entrust with this critical piece of the CRISPR workflow.
Why Turn to Multispan?
- 100% success rate for generation of quality clonal stable cell lines for >500 targets
- Cell engineering expertise and rigorous QC for rapid production of clonally derived stable cell lines
- Collaborations with leading providers of CRISPR nuclease tools and technologies enabling optimum upstream platform selection, gRNA design and optimization, and analysis
- Clonal selection using functional read outs resulting in engineered cells ready for incorporation into high throughput screening assays
CRISPR Cell Engineering Solutions That Propel Drug Discovery
- Stable pool or clonally derived cell line generation
- Clone Screening:
- Heterozygous or homozygous stable cell line selection by function
- Selection against parental or orthogonal cell lines
- Cell line validation using real-time PCR and FACS
- Expansion and banking of CRISPR stable cell lines, including production of assay ready, division-arrested cells
- Seamless integration into Multispan’s full assay development, screening and profiling services for accelerated drug discovery
- CRISPR design and molecular cloning can be seamlessly integrated within projects by working with our preferred collaborators IDT and Harvard.
It’s through the creation of quality stable cell lines that the power of CRISPR engineering is harnessed. Trust Multispan’s expertise in cell line engineering and breadth of services to bring your CRISPR initiatives alive. Your success is our priority.