Custom Drug Discovery Services & High Throughput Screening
Fast turn-around, 384-well format cell-based, phenotypic, and binding assays
To harness the power of fully integrated cell engineering, assay development and high throughput screening capabilities at Multispan, we offer compound screening and profiling services.
Details inevitably get lost in the transfer of cell lines, reagents, and protocols of cell culture and HTS assay from site to site. As a result, redundant learning, relearning, and troubleshooting leads to significant wasted time and cost. The elimination of these transfer steps by performing cell engineering, assay development and compound screening in one site argues strongly for time and cost-efficiency. Multispan excels at all three! Our 1000 HTS-ready MULTISCREENTM GPCR cell-based assays can further help shorten the timeline by jumpstarting the screens. It is also noteworthy, the newly developed MULTISCREENTM β-Arrestin Sensor makes HTS for orphan GPCRs (oGPCRs) possible for the first time in their untagged native form.
Multispan has extensive experience in screening and profiling compounds in a variety of HTS cell-based assay formats including ELISA, cell-based ELISA, reporter, binding, 2nd messenger, flow cytometry (FACS) assays in fluorescence, chemiluminescence, TR-FRET, absorbance, and radioisotope readouts. Depending on the biology of interest, we have screened compounds in agonist, partial agonist, inverse agonist, antagonist, partial antagonist, positive allosteric modulator (PAM) and negative allosteric modulator (NAM) modes. The majority of the screens are performed in 384-well microplates formats.
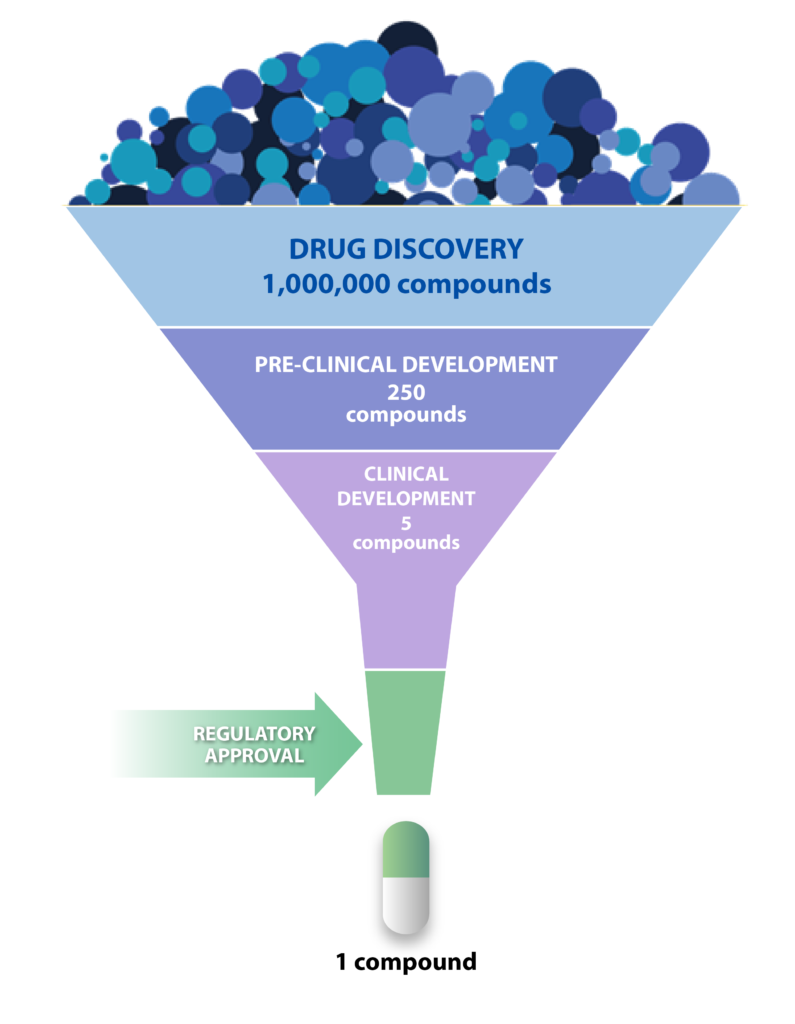
In our compound profiling assay services and high-throughput screening services, quality and speed are our priority.
We make sure not only our cell-based HTS assays run with expected pharmacology determined by control ligands, but also the Z-factor or Z’ meets the HTS requirement at 0.5, which we exceed most of the time. Such controls are included in each 384-well microplate. In the very rare event that data from a control compound didn’t run as expected or consistently, we would repeat the whole plate with no questions asked. The entire dataset from control and testing compounds is shared in raw and analyzed formats. It’s of the utmost importance that we offer 100% transparency in data sharing so that our clients can draw the right conclusions based on complete facts.
To match the uncompromising quality, our HTS and compound profiling services also come with exceptionally fast turnaround.
- HTS of 100,000 compound library and hit confirmation ⇒ in 1 month
- Weekly SAR lead optimization primary assay ⇒ in 1 day
- Monthly SAR lead optimization secondary assays ⇒ in 1-5 days
- 32-GPCR safety functional assay panel screening ⇒ in 5 days
- 231-GPCR panel screening ⇒ 4 weeks
For Example...
We were asked by a virtual biotech company to help develop an HTS screening funnel for a nuclear hormone receptor. We started by studying the literature and brainstorming with academic KOLs to gain thorough understanding of the target biology and potential assays that can be developed. We developed 2 sets of assays in tandem: a reporter gene assay as the primary screen and a phenotypic assay using endogenous readout as the translational screen. Both assays were developed in human and rodent species, the latter matching the animal model that will be used later in the program. We conducted routine screens in the primary assay every other week with 1-day turnaround whereas the translational assay every other month with also 1-day turnaround. The program successfully progressed until a lead candidate was selected for IND.
Our combined expertise in cell engineering, assay development and high throughput screening has allowed us to build a consistent track record in high performance at high efficiency over nearly 2 decades.
Our compound profiling and high throughput screening services are aimed to bring extraordinary time and cost savings to our clients in drug discovery from target to IND.
FAQs
High-throughput screening or HTS is a drug development technique used to rapidly evaluate the biological activity of a large number of compounds and targets. It is an essential part of discovering potential therepeutic compounds, known as lead candidates.
Virtual screening utilizes computer simulations to model compound interactions with a target, saving time and cost in the early stages of drug discovery where a subset of potential compounds must be identified for further validation in biological assays. HTS, on the other hand, requires more resources and time to physically test interactions between compounds and targets, but provides tangible data about compound activity. Virtual screening can be used to enhance HTS by narrowing down the scale of the actual campaign.
Microplates or microtiter plates are commonly used in HTS. While 384-well and 1536-well plates are ideal for greater throughput due to a higher density, 96-well plates are also amenable for smaller screening operations. Microfluidics systems are also used when small volumes of fluids and efficient reagent use are required.
A high-throughput screening campaign usually consists of these main steps: target identification, assay development, compound screening, analysis of results and hit selection.
Depending on the capabilities of the screening facilities, readiness of the bioassay, and the size of the compound library, HTS can take less than a week (for a small library with thousands of compounds) or a few months (for a large library with hundreds of thousands to millions of compounds). The overall HTS campaign duration, however, will vary significantly depending on the scope of the project.
Additional Resources:
Article: High Throughput Screening Done Right
Article: Hit-to-Lead and Lead Optimization In Vitro Pharmacology Support
Article: 32-GPCR Safety Functional Assay Panel for Routine Off-Target Compound Profiling
Article: Strategies and Tools to Develop a Better GPCR-Targeted Therapeutic
Article: Assay Orphan GPCRs in High Throughput with MULTISCREENTM β-Arrestin Sensor Technology
