Product Information
Catalog Number: C1243
Lot Number: C1243a-030121
Quantity: 1 vial (2 x 106) frozen cells
Freeze Medium: Cellbanker 2
Host cell: HEK293T
Transfection: Expression vector containing full-length human FPR1 cDNA (GenBank Accession Number NM_002029.3) with FLAG tag sequence at N-terminus.
Recommended Storage: Liquid nitrogen upon receiving
Propagation Medium: DMEM, 10% FBS, 1 μg/mL puromycin
Stability: Stable for a minimum of 2 months in continuous culture
Data Sheet
Background: The gene FPR1 encodes the formylpeptide receptor (FPR), which is a G-protein-coupled receptor that mediates chemotaxis of phagocytic leukocytes induced by bacterial peptide N-formyl-methionyl-leucyl-phenylalanine (fMLP). Agonist binding to FPR in phagocytic leukocytes leads to the activation of phosphatidylinositol 3-kinase (PI3K), mitogen-activated protein kinases (MAPKs), and the transcription factor nuclear factor (NF)-kB via heterotrimeric Gαi proteins. FPR is involved in host defense against bacterial infection and in the clearance of damaged cells. Recently a large number of non-formylated peptide ligands for FPR have now been identified. Some of the new ligands (e.g. Ac1–26 from annexin) are endogenous in origin, and some come from pathogens that are associated with human diseases such as HIV, which have suggested novel roles for this receptor in the regulation of acute and chronic inflammation as well as host responses during HIV-1 infection.
Application: Functional assays
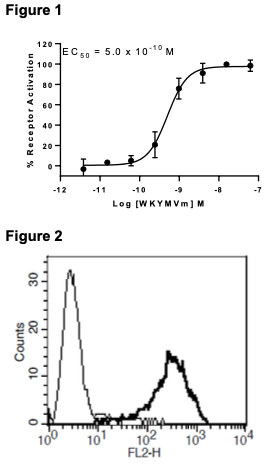
Figure 1. Dose-dependent inhibition of forskolin-stimulated intracellular cAMP accumulation upon treatment with ligand, measured with Multiscreen™ TR-FRET cAMP 1.0 No Wash Assay Kit (Multispan MSCM01). Figure 2. Receptor expression on cell surface measured by flow cytometry (FACS) using an anti-FLAG antibody. Thin line: parental cells; thick line: receptor-expressing cells.
References:
Le et al. (2002) Formyl-peptide receptors revisited. Trends Immunol 23:541-548.
Torres and Ye (1996) Activation of the mitogen-activated protein kinase pathway by fMet-leu-Phe in the absence of Lyn and tyrosine phosphorylation of SHC in transfected cells. J Biol Chem 271:13244-13249.
